
Metalloids have some properties of metals and some properties of nonmetals. It’s helpful to know the difference between metals and nonmetals in order to identify metalloids. Readily gain or share valence electrons.Usually have 4-8 electrons in their outer shell.May be solids, liquids or gases at room temperature.Poor conductors of heat and electricity. 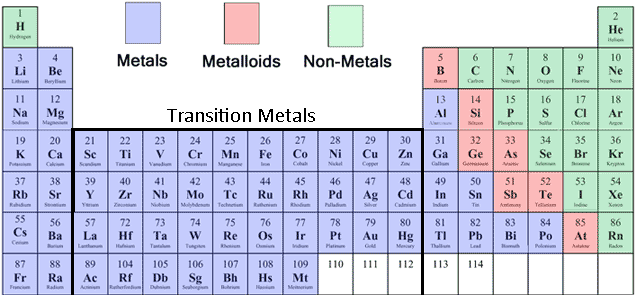
Not lustrous (dull appearance, sometimes colorless).The nonmetals are a diverse collection of elements, but they share some common properties. While most metals are solid at room temperature, there nonmetallic solids, liquids, and gases. Nonmetals include the nonmetal, halogen, and noble gas groups on the periodic table. Corrode easily (e.g., damaged by oxidation such as tarnish or rust).Have 1-3 electrons in the outer shell of each metal atom and lose electrons readily.Metals are sonorous or make a bell-like sound when struck.Opaque as a thin sheet (can’t see through metals).Usually solid at room temperature (an exception is mercury).Good conductors of heat and electricity.One common property is metallic character, which means atoms of metals readily lose electrons. 
But, they share other properties in common with each other. Most metals are easy to recognize by their shiny, metallic appearance. Because so many elements are metals, they are subdivided into smaller groups, including the alkali metals, alkaline earth metals, transition metals, post-transition or basic metals, lanthanides, and actinides. MetalsĪbout 75% of elements on the periodic table are metals. Solid, liquid, or gas at room temperatureĬomparison of the chemical and physical properties of metals and nonmetals. Metals, metalloids, and nonmetals on the periodic table Metals Here is a look at the difference between metals and nonmetals. Metalloids occur in zig-zag line on the table, separating metals and nonmetals. Nonmetals are on the right side of the table. Metals are on the left side of the periodic table. Elements with some characteristics of metals and some of nonmetals are metalloids. Rather, they fall somewhere in the middle regarding their appearance and other properties.This entry was posted on Augby Anne Helmenstine (updated on August 9, 2021)Įlements may be classified as metals, nonmetals, or metalloids. They aren’t particularly lustrous, nor are they are particularly dull. Metalloids are unique because their properties fall somewhere between those of metal and nonmetal elements. In addition to metals and nonmetals, there are also metalloids on the periodic table. Additionally, nonmetal elements have a dull appearance, which is in stark contrast to the lustrous appearance exhibited by metal elements. They aren’t able to pass electricity or heat as well as metal elements. Nonmetal elements have poor electrical, as well as thermal, conductivity. Metal elements are also malleable and ductile, allowing them to be “worked.” In other words, it takes more heat for a metal element to melt than it does for a nonmetal element to melt. Furthermore, metal elements have a higher melting point than that of nonmetal elements. They are able to pass electricity with minimal restriction, making them ideal for use in wires and cables. While each element on the periodic table has its own unique properties, all metal elements generally share some common characteristics, and all nonmetal elements also share some common characteristics.įor starters, metal elements have high electrical conductivity properties. Now that you know the basic definitions of metal and nonmetal elements, you might be wondering how they differ. Differences Between Metal and Nonmetal Elements Nonmetal elements are rarer than metal elements, but there are still plenty of them available on the periodic chart. Examples of nonmetal elements include hydrogen, helium, nitrogen, oxygen, fluorine, chlorine, sulfur, argon, xenon, radon and phosphorus. Nonmetal elements don’t form positive ions, nor do they possess metallic bonds. Examples of metal elements include iron, copper, silver, mercury, lead, aluminum, gold, platinum, zinc, nickel and tin.Ī nonmetal element, on the other hand, is an element that lacks the characteristics of its metal counterparts. Most elements on the periodic table are metals. What Is a Metal Element?īy definition, a metal element is an element that form positive ions and has metallic bonds. Aside from their placement on the periodic table, though, there are several key differences between metal and nonmetal elements. The former are found on the left side of the periodic table, whereas the latter are found on the right side of the periodic table. There are currently 118 known elements on the periodic table, many of which are classified as either a metal or nonmetal.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
